High-Throughput physiology-based stress response phenotyping: Advantages, applications and prospective in horticultural plants
A B S T R A C T
Phenomics is a new branch of science that provides high-throughput quantification of plant and animal traits at systems level.
The last decade has witnessed great successes in high-throughput phenotyping of numerous morphological traits, yet major challenges still exist in precise phenotyping of physiological traits such as transpiration and photosynthesis. Due to the highly dynamic nature of physiological traits in responses to the environment, appropriate selection criteria and efficient screening systems at the physiological level for abiotic stress tolerance have been largely absent in plants. In this review, the current status of phenomics techniques was briefly summarized in horticultural plants.
Specifically, the emerging field of high-throughput physiology-based phenotyping, which is referred to as “physiolomics”, for drought stress responses was highlighted. In addition to analyzing the advantages of physiology-based phenotyping over morphology-based approaches, recent examples that applied high-throughput physiological phenotyping to model and non-model horticultural plants were revisited and discussed.
Based on the collective findings, we propose that high-throughput, non-destructive, and automatic physiological assays can and should be used as routine methods for phenotyping stress response traits in horticultural plants.
1. Introduction
Over the past few decades, considerable progress has been made in “omics” technologies ( Urano et al., 2010 ; Li, 2013 ). Since the whole genome sequence of Arabidopsis thaliana was assem- bled ( Initiative, 2000 ), genomics has led the way in technical “omics” advancements along with other related technologies, such as transcriptomics and epigenomics. Moreover, the appli- cation of high-resolution mass spectrometry technology has fostered the development of proteomics and metabolomics ( Aebersold and Mann, 2003 ; Wang and Bodovitz, 2010 ).
While these technologies have been valuable in uncovering the molec- ular mechanisms of plant development and interactions with the environment, a fundamental understanding of plant biology requires high-quality, multi-faceted phenotypic data. Despite the emergence of the “phenome” in the 1990s ( Schork, 1997 ; Siebner et al., 2009 ), which refers to a comprehensive set of phenotypes in a living organism, the utilization of phenomics in plant science has lagged behind the genomics technology until several key supportive technologies/tools became available ( Bilder et al., 2009 ; Houle et al., 2010 ), including high-throughput imaging, high-precision sensor, automatic control, and infor- mation processing systems, as well as data decoding software ( Mahner and Kary, 1997 ; Varki et al., 1998 ; Gerlai, 2002 ; Schilling et al., 2010 ).
Subsequently, phenomics has been widely applied to analyze phenotyping plants and animals at the cellular, organ, individual, and population levels, providing abundant, comprehensive, and reproducible data ( Finkel, 2009 ).
Due to the weaknesses of traditional phenotyping meth- ods such as low-throughput, labor cost and strong subjectivity ( Furbank, 2009 ), so they are becoming less powerful in meeting the need of comprehensive analysis of the phenome-genome- environment relationships.
Phenomics is a powerful approach that quantitatively measures plant morphological, biochemical, and physiological characteristics under genotype-environment interactions. Equipped with large-scale, non-destructive, and au- tomated phenotyping facilities, plant phenomics systems pro- vide dynamic and comprehensive phenotypic data for traits of in- terest ( Finkel, 2009 ). These data are key for biological research and trait-based plant breeding or pre-breeding ( Furbank and Tester, 2011 ). Generally, current phenomics platforms, according to their targeted traits, are divided into 2 categories, morphology-based and physiology-based phenomics platforms. The former acquires morphological parameters, such as plant height, stem diameter, leaf area, leaf angle, stem length, and plant spacing.
The latter ac- quires physiological indexes, including chlorophyll fluorescence, transpiration rate, leaf water content, root influx and efflux, and stomata conductance. Irrespective of morphological and physio- logical traits, phenomics requires that these traits be monitored simultaneously in multiple plants in order to obtain homoge- neous data for comparison ( Close, 2011 ). After obtaining these morphological and physiological data, the information can be in- tegrated with genome-wide DNA genotypic data for the identifi- cation of genetic determinates of specific traits via genetic link- age mapping (for pedigree populations, such as F 2 , BC 1 F 1 , and recombinant inbred lines) or association mapping (for natural populations). Phenomics applications have moved beyond staple food crops (e.g., rice, wheat, and maize) to more economic plants, including horticultural plants (e.g., tomato, beans, and cucumber) (Xu et al., 2015 ; Halperin et al., 2017 ; Hui et al., 2018 ; Zhou et al., 2020 ).
In this paper, the current status of phenomics techniques in horticultural plants was briefly reviewed. The emerging field of high-throughput physiology-based phenotyping is highlighted and the term “physiolomics” was coined to specifically refer to the “omics” of physiology. By showcasing its applications in model horticulture plants tomato and non-model horticulture plants yardlong bean (vegetable cowpea) and pepper, we appeal that high-throughput, non-destructive, and automatic physiolog- ical assays can and should be used as routine technological meth- ods for stress response traits phenotyping in horticultural plants.
2. Morphology-based stress responses phenotyping Morphological changes are relatively easy to detect and monitor.
Traditional stress response phenotyping, either by manual measurements or using imaging tools, relies more upon morphological indicators to quantify stress responses. Due to the advancement of optical imaging and remote sensing technologies, morphological phenotypes of several plants can be measured automatically, simultaneously, and continuously ( Parent et al., 2015 ).
Current high-throughput scanning systems use dozens to hundreds of sensors to measure a variety of mor- phological traits, such as plant height, canopy size, leaf area, leaf greenness, branch angle, disease spot size, and plant wilt degree. By combining these data, one can comprehensively characterize important traits, such as plant architecture, nutritional status, drought tolerance, and disease resistance. To date, these technologies have been applied to many horti- cultural plants. Hurtado et al. (2013) evaluated fruit shape traits in 21 eggplant accessions from 4 varieties using the Tomato An- alyzer image tool. A total of 23 fruit shape parameters were eval- uated for each accession, including fruit shape index, blocki- ness, homogeneity, proximal fruit end shape, asymmetry, inter- nal eccentricity, and slenderness.
Hui et al. (2018) obtained the three-dimensional (3D) canopy of cucumber, pepper, and egg- plant based on multi-view stereo (MVS) around the plant canopy. Using this approach, the parameters of leaf length, leaf width, leaf area, plant height, and maximum canopy width were ex- tracted and calculated which demonstrated that the dynamic capture of highly accurate 3D canopy structures was a poten- tial approach for assisted breeding and field management prac- tices. Jaradat (2018) identified phenotypic data for agronomic and stress response traits using the Crop Circle ACS-470 multi- spectral crop canopy sensor platform, including low-temperature tolerance during germination, early seedling growth, flowering, and maturity, and biomass under field conditions. These param- eters were used to select high-yield Brassica napus varieties suit- able for short-term growth in the Midwestern United States.
Using the Scanalyzer 3D large-scale imaging platform, Laxman et al. (2018) digitally quantified tomato biomass and detected signifi- cant linear relationships between the projected shoot area (PSA), plant fresh mass, and plant digital biomass. Moreover, Bernotas et al. (2019) argued that a 3D morphological phenotyping system based on photometer stereoscopic (PS) imaging would be invalu- able for phenotyping circadian rhythms in eudicot species, such as tomato, cabbage, and oilseed rape, by tracking the rhythmic movements of cotyledons at the seedling stage.
3. Physiology-based stress response phenotyping Despite being powerful and widely use, morphology-based phenomics assays still have many limitations.
One drawback is the slow expression of morphological traits, usually over the course of days to weeks after treatment. Additionally, due to the complexity of plant architecture and field conditions, mechanical morphological phenotyping can be inaccurate or even erroneous ( Poorter et al., 2016 ; Fernandez et al., 2017 ; Fischer and Rebet- zke, 2018 ).
Physiology-based phenotyping provides more sensi- tive measurements of plant phenotypes, making it a better fit for plant-environment interaction analysis ( Earl, 2003 ; Pereyra-Irujo et al., 2012 ; Halperin et al., 2017 ). Among the many environmental cues, water stress due to soil drought is a global threat to agriculture. Under current climate deterioration trends, crop yield loss because of drought stress will become more frequent.
Plant drought responses can be measured by morphological traits, such as the leaf yellowing index and wilting degree. However, from an agronomic per- spective, drought tolerance should always be assessed based on minimum yield losses compared with non-stressed controls, which is closely related to plant growth recovery after the removal or alleviation of stress ( Hatfield and Walthall, 2015 ).
Drought tolerance is a complex quantitative trait with many determinants and low inheritability. It is difficult to perform phenotypic analysis using morphological measurements and should be conventionally evaluated at the final stage of a plant’s lifecycle by calculating yield loss ( Negin and Moshelion, 2017 ). Previous studies uncovered linear relationships between the quantities of intercepted solar radiation, water, CO 2 , and dry matter production in crops ( Dewar, 1996 ; Sinclair and Muchow, 1999 ; Hall, 2001 ), which provided a means for physiologically predicting crop yield loss. However, one challenge of physiology- based stress phenotyping in field conditions is the degree of stress, which is closely related to dynamic soil-atmosphere conditions ( Asfaw et al., 2012 ). Accordingly, the parameters used for quantifying drought tolerance are also dynamic.
Recently, there has been a breakthrough in the technolo- gies used for physiology-based stress phenotyping. For exam- ple, a variety of detecting platforms and statistical methods used to obtain physiological trait data continuously and in a high- throughput manner ( Ghanem et al., 2015 ). Weighing lysimeters are widely used for tracking changes in plant weight, which allows for the monitoring of plant water-loss rates and water use efficiency (WUE), as well as the simulation of drought stress ( Earl, 2003 ; Pereyra-Irujo et al., 2012 ; Vera-Repullo et al., 2015 ).
Similarly, a non-destructive, automated, high-throughput system (PlantArray) was developed for water and biostimulant responses pheno- typing based on physiological traits ( Halperin et al., 2017 ; Dalal et al., 2019 ).Theoretically, the system is extendible to other types of abiotic stress, including salt, low or high temperature, and heavy metals. This platform monitors many physiological traits, includ- ing photosynthesis and transpiration rates, which are key factors of maintaining crop yield under stress conditions. These physio- logical traits are referred to as “functional traits” or “quantitative physiological traits” (QPTs) ( Violle et al., 2007 ). Accordingly, the high-throughput physiolomics platform allows for the functional physiological phenotyping (FPP) of plants.
Through the simul taneous monitoring of the environment (soil-atmosphere) and QPTs of hundreds of plants, the stress responses among several germplasm lines can be compared ( Wallach et al., 2010 ; Yin and Struik, 2016 ). By comparing the dynamic performance of each plant with respect to the whole population, plants that perform well under a specific stress scenario can be easily selected. More- over, the underlying physiological mechanisms of genotypic dif- ferences on the stress responses of plants can also be uncovered. Due to the higher sensitivity of physiological responses than mor- phological changes to the environment, FPP can more effectively detect subtle or transient stress responses ( Negin and Moshelion, 2017 ).
Exemplary studies that employed the physiolomics plat- form for FPP in horticultural plants were discussed below.

Fig. 1 High-throughput physiological assay systems for stress-response phenotyping A. Lysimetric system used for tomato plants (photograph from Halperin et al., 2017). B. Lysimetric system used for yardlong bean plants (photograph from Xu et al., 2015). C. An updated lysimetric system that compensated for temperature and light in Huai’an, Jiangsu Province, China (photograph captured in October 2019).
4. Applications of physiolomics assays in horticultural plants
4.1. Applications in the model horticulture plant tomato With a relatively small genome, short growth cycle, and well- established transgenic systems, the tomato is a fleshy model horticulture plant. The study of functional genomics and phe- nomics in tomato has become a hotspot of horticulture research.
Halperin et al. (2017) and Nir et al. (2017) used the physiolomics system to record the profiles of physiological responses in an ar- ray of plants under normal, water-stressed, and recovery con- ditions in a real-time manner ( Fig. 1 , A). The system allowed for the simultaneous and continuous monitoring of whole-plant transpiration, biomass gain, stomatal conductance, and root flux with specifically designed algorithms. Compared with the con- ventional gasexchange tools, this physiological assay possesses major advantages in measuring several plants at a higher tem- poral resolution, as well as the comparative examination and graded ranking of different physiological characteristics, including growth rate, WUE, mid-day transpiration level ( E ), canopy stomatal conductance ( G sc ), and the drought-resistance index (DRI). Based on these QPT data, plant resilience can be easily as- sessed, and the genotypes exhibiting the fastest and most thor- ough recovery upon re-watering after drought stress can be se- lected. Thus, this physiological system links plant functional ge- nomics and modern breeding technology.
4.2. Applications in the non-model horticultural plants cowpea and pepper Each plant species or cultivar uses its own pathway to bal- ance its water status, which has important implications on plant growth behaviors and yield penalty under stress conditions.
Ac- cording to the physiological characteristics of whole-plant water relations, plants can be divided into the isohydric and anisohy- dric types ( Tardieu and Simonneau, 1998 ). Isohydric plants (e.g., cactus) prioritize water conservation when faced with drought stress by quickly closing their stomata to prevent water loss, which consequently retards growth.
In contrast, anisohydric plants (e.g., sunflower) maintain relatively high stomatal con- ductance ( G S ) and CO 2 assimilation rates ( A N ) under drought stress, specifically under light to moderate drought conditions, in order to gain biomass ( Tardieu and Simonneau, 1998 ; Aharon et al., 2003 ; Uehlein et al., 2003 ). Originating from the arid re- gion of West Africa, cowpea exhibited excellent drought toler- ance and broad intra-species variation in this trait ( Ehlers and Hall, 1997 ; Muchero et al., 2008 ). Previous studies revealed 2 differ- ent types of drought-tolerant responses in cowpea, types I and II ( Watanabe et al., 1997 ; Muchero et al., 2008 ; Agbicodo et al., 2009 ), which corresponded to isohydric/anisohydric behaviors, respectively, rendering cowpea as a promising new model system for studying water relations, including isohydric/anisohydric water adjustment mechanisms.
Using a similar platform, Xu et al. (2015) optimized the growth conditions and experimental procedures for the physiological stress response assays of yardlong bean (vegetable cowpea), which included ambient temperate ranges, lights, water supply modes, physiological parameters, and curation ( Fig. 1 , B). The sta- bility and reliability of the platform were verified through the continuous monitoring of 4 plant varieties B118, B128, B253 and B47 for 15 d Genotypic differences in water regulation strategies among the 4 varieties were well-demonstrated. The results re- vealed significant differences in WUE, the maximum transpira- tion ( E max ), daily transpiration, and growth inhibition degree un- der drought stress among the 4 varieties ( Fig. 2 , A and B). The semi-dwarf variety, B47, exhibited a slow growth rate and moder- ate WUE with a less drastic loss of growth potential in response to drought treatment, while an indeterminate-type variety, B128, exhibited the fastest growth rate and the highest WUE under well-watered conditions, as well as the greatest loss of growth potential under drought treatment.
These two materials are considered to behave the characteristics of isohydric and anisohy- dric types to some degree, respectively. Moreover, by aligning the dynamic soil water content ( θ) data with the transpiration rate trajectories, the authors were able to calculate the critical soil water content ( θcr ), which is the key secondary parameter de- noting when a plant starts to close its stomata, for each variety ( Fig. 2 , A). Moreover, the whole-plant daily transpiration and whole plant weight among genotypes were compared over the course of pre-treatment, stress, and recovery phases with 4 plant varieties G034, G047, G356 and TZ30 for 27 d ( Fig. 2 , C and D). Un- der drought stress condition, the growth inhibition degree of G047 was significantly higher than TZ30 ( Fig. 2 , C and D).
These quantitative water budgeting parameters are valuable for the breeding and molecular dissection of water regulation behaviors in this species. In addition to drought, Dalal et al. (2019) investigated the effects of different biostimulants on pepper plants under dif- ferent irrigation regimes using a similar system with minor technical modifications. The effects of 2 biostimulants, ICL- SW and ICL-NewFo1, on plant daily transpiration, volumetric water content (VWC), biomass, and WUE were quantified and compared using the system; fruit number under well-irrigated and drought stress scenarios was also measured. Compared with the control, biostimulant application improved the overall transpiration and biomass, which was considered as an alterna- tive approach to increase crop productivity. Additionally, 2 new resilience-quantifying-related traits were proposed, transpiration recovery rate and night water reabsorption, which can also be used for phenotype identification by the system. This study demonstrated that physiology-based phenotyping due to its dynamic, accurate, and high-resolution nature can effectively detect small or short-term physiological changes, rendering it valuable for predicting yield fluctuations under favorable or unfavorable conditions.
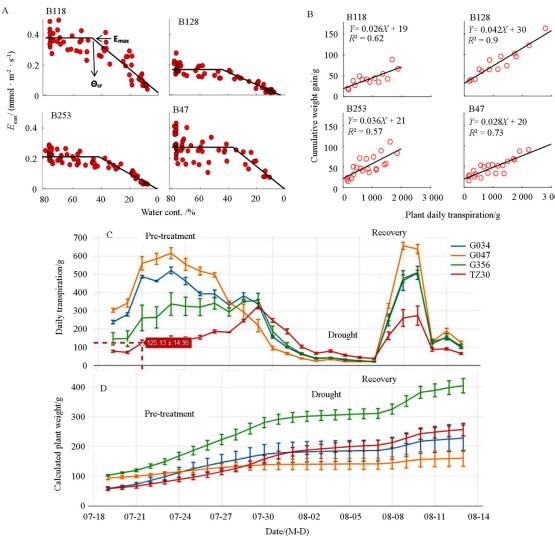
Fig. 2 An optimized physiological system and the experimental results for QPT measurements in yardlong bean, a non-model vegetable legume A. Noon-day transpirations of 4 Chinese yardlong bean varieties (B118, B128, B253 and B47) plotted against soil water contents. For each variety, the critical soil water content (θcr) when a plant started to close its stomata was clearly observed and indicated as the turning point. Emax is the daily maximum transpiration rate normalized to plant weight (Xu et al., 2015). B. Plant water use efficiency (WUE) in the 4 varieties was calculated as the ratio between plant weight gain and the amount of water that was transpired. The WUE of each variety was determined by fitting a linear curve of the cumulative plant weight gain during the pretreatment stage vs. cumulative water transpiration (Xu et al., 2015). C. Whole-plant daily transpiration of another 4 Chinese yardlong bean varieties (G034, G047, G356 and TZ30) throughout the experimental period (27 d, unpublished data). D. Whole-plant weight throughout the experimental period (27 d, unpublished data).
5. Future prospective Both morphology- and physiology-based phenotyping meth- ods are rapidly deployed in horticulture plant research.
For the former, there is a need to develop cost-effective and adaptable infrastructure in the near future to analyze multi-dimensional phenotypes. More sophisticated and automated workflows are also required to implement proper checkpoints at different stages of the phenotyping process to reduce the risk of data quality deterioration and the omission of interesting phenotypes ( Rahaman et al., 2015 ; Araus et al., 2018 ). Given the fast and sensitive responses of plants to the environment at the physi- ological level when compared with the morphological level, the latter (physiology-based parameters) is directly and promptly relate to the regulation of DNA, RNA, and proteins.
Therefore, there is a potential for physiolomics to enable the systematic analysis of phenotypic data with other “omics” data, such as genomics, transcriptomics, metabolomics, and epigenomics data, to better relate plant stress responses to their environ- ments. Genome-wide association studies (GWAS) have also been widely used to identify specific genome regions controlling key physiological traits that confer to stress tolerance. A challenge in the near future is the efficient dissection of genetic components that control dynamic physiological changes over time, given the increasingly massive amount of phenotypic data. New statistical ramework, such as “functional mapping,” which is a general statistical mapping framework used to characterize quantitative trait loci (QTLs) of a complex dynamic trait in a single step, could be valuable in overcoming this challenge and ultimately enable the interpretation of big data acquired from physiolomics facilities ( Wu and Lin, 2006 ; Li and Sillanpää, 2013 ). With the rapid increase of physiological system applications in horticulture plant research, further technical advancements are expected to be achieved in the following areas: first, more convenient and user-friendly operation systems should be de- veloped for plant physiologists, geneticists, and breeders alike, of whom the latter two usually lack professional plant physiology knowledge; second, more complex, secondary physiological parameters should be calculated from many primary parameters that are measurable by the system.
This category could include the relative water content (RWC) of leaves ( Anjum et al., 2011 ; Kalariya et al., 2015 ; Tanentzap et al., 2015 ), as well as leaf ( leaf ) and root water ( root ) potentials. Acquiring a more comprehensive set of parameters will deepen our understanding of the whole-plant water budgeting process of plants in response to changing environmental conditions. For example, due to dynamic soil-atmosphere parameters, which are measured along with plant physiological traits in physiological assays, transpiration rates and other key stress responses in different plant varieties can be compared based on soil moisture contents rather than the duration of stress treatment used in traditional methods. Such advancements will provide more physiolog- ically meaningful insights into the genotypic differences in horticultural plants. Acknowledgements The authors wish to thank Menachem Moshelion for useful discussions. This work is supported by National Natural Science Foundation (NSFC) of China (Grant No. 31772299 ), NSFC-Israeli Science Foundation (ISF) joint project (Grant No. 31861143044 ) and National Program for Support of Top-Notch Young Profes- sionals (to P.X.).
